Resistell’s first patients recruited for international clinical study
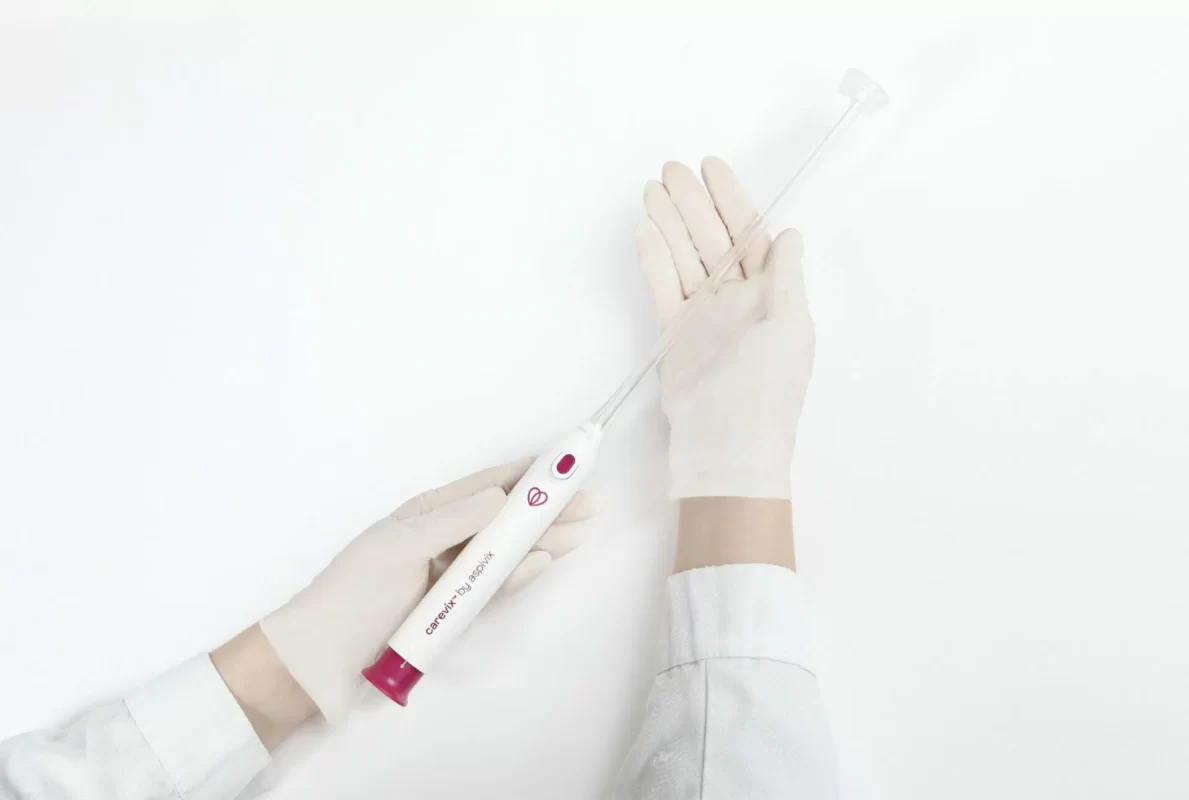
HEMEX is proud to announce that our portfolio company ASPIVIX has received 510(k) clearance of Carevix™ from the U.S. Food and Drug Administration (FDA). ASPIVIX’s novel Cervical Stabilizer will allow millions of women across the USA access to significantly less painful treatments and IUD insertions.
New Year’s resolutions can be a powerful way to make positive changes in life and achieve goals.
Increasing patients’ safety and care in research is vital when preparing and conducting your study. From a HEMEX perspective, we are tackling in this article four important points you should consider during the process.
We are thrilled to share that HEMEX is part of the investment consortium financing the series B1 of hemotune AG. At HEMEX, we look forward to support hemotune on the journey to making the HemoSystem available to patients globally.